 , Hye Kyung Chang, Min Jeong Cho, Yong Hoon Cho, Soo Jin Na Choi, Yoon Mi Choi, Jae Hee Chung, Sang Young Chung, Jeong Hong, Seok Joo Han, Yeon Jun Jeong, Eunyoung Jung, Kyuhwan Jung, Dae Youn Kim, Hae-Young Kim, Hyun-Young Kim, Ki Hoon Kim, Sang Youn Kim, Seong Chul Kim, Seong Min Kim, Soo-Hong Kim, Jong-In Lee, Myung-Duk Lee, Nam-Hyuk Lee, Suk-Koo Lee, So Hyun Nam, Jin Young Park, Kwi-Won Park, Tae-Jin Park, Jeong-Meen Seo, Jae Ho Shin, Jiyoung Sul
, Hye Kyung Chang, Min Jeong Cho, Yong Hoon Cho, Soo Jin Na Choi, Yoon Mi Choi, Jae Hee Chung, Sang Young Chung, Jeong Hong, Seok Joo Han, Yeon Jun Jeong, Eunyoung Jung, Kyuhwan Jung, Dae Youn Kim, Hae-Young Kim, Hyun-Young Kim, Ki Hoon Kim, Sang Youn Kim, Seong Chul Kim, Seong Min Kim, Soo-Hong Kim, Jong-In Lee, Myung-Duk Lee, Nam-Hyuk Lee, Suk-Koo Lee, So Hyun Nam, Jin Young Park, Kwi-Won Park, Tae-Jin Park, Jeong-Meen Seo, Jae Ho Shin, Jiyoung SulThe Korean Association of Pediatric Surgeons, Seoul, Korea.
Copyright © 2019 Korean Association of Pediatric Surgeons
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.

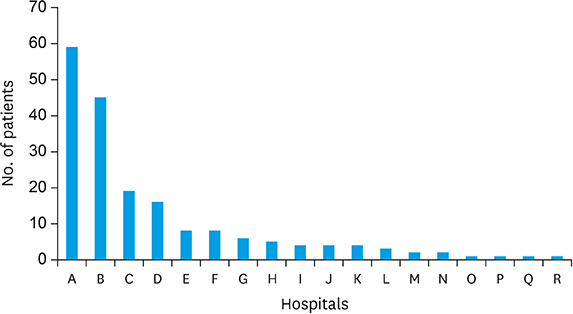
| Year and topic | |||
|---|---|---|---|
| 1991 | Current situation in Korean pediatric surgery | 2005a) | Necrotizing enterocolitis |
| 1992 | Inguinal hernia | 2006a) | Acute appendicitis |
| 1993 | Hirschsprung disease | 2007 | Prospect of pediatric surgery |
| 1994 | Anorectal malformation | 2008 | Inguinal hernia |
| 1995a) | Esophageal atresia and tracheoesophageal fistula | 2009 | Hirschsprung disease |
| 1996a) | Branchial anomalies | 2010a) | Intestinal atresia |
| 1997a) | Infantile hypertrophic pyloric stenosis | 2011a) | Biliary atresia |
| 1998a) | Intestinal atresia | 2012 | Statistics of pediatric surgery disease |
| 1999a) | Anorectal malformations | 2013a) | Minimally invasive surgery |
| 2000a) | Index cases in pediatric surgery | 2014a) | Newborns surgery with congenital anomalies |
| 2001a) | Biliary atresia | 2015a) | Neonate congenital Bochdalek hernia |
| 2002a) | Choledochal cyst | 2016 | Esophageal atresia with tracheoesophageal fistula |
| 2003a) | Congenital posterolateral diaphragmatic hernia | 2017 | Choledochal cyst |
| 2004 | Trend of pediatric surgery disease | 2018a) | Sacrococcygeal teratoma |
a)These studies were published in Advances in Pediatric Surgery.
| Characteristic | Value | |
|---|---|---|
| Sex (M:F) | 1:2.71 (51:138) | |
| Gestational age (wk, n=176) | 37.9±2.8 | |
| Birth weight (kg, n=179) | 3.18±0.52 (0.97–4.71) | |
| Mode of delivery | ||
| Normal spontaneous vaginal delivery | 76 (40.2) | |
| C-section | 100 (52.9) | |
| Unknown | 23 | |
| Accompanied malformation | 29/189a) (15.3) | |
| Cardiovascular | 8 | |
| Gastrointestinal | 5 | |
| Genitourinary | 4 | |
| Musculoskeletal | 4 | |
| Chromosomal | 2 | |
| Other | 6 | |
| Currarino syndrome | 15 | |
| Age at time of surgery | ||
| <29 day | 137 | |
| 29 day–2 mo | 6 | |
| 2–3 mo | 7 | |
| 3–12 mo | 12 | |
| >12 mo | 27 | |
Values are presented as mean±standard deviation or number (%).
a)Including multiple selection.
| Characteristic | Total | Neonate | Old-age | |
|---|---|---|---|---|
| Clinical presentationa) | (n=182) | (n=132) | (n=26) | |
| Abdominal pain/distension | 13 (7.1) | 5 (3.8) | 2 (7.7) | |
| Mass | 145 (79.7) | 120 (90.9) | 11 (42.3) | |
| Constipation | 10 (5.5) | 0 | 5 (19.2) | |
| No symptoms | 9 (4.9) | 7 (5.3) | 1 (3.8) | |
| Other | 14 (7.7) | 1 (0.8) | 9 (34.6) | |
| Prenatal diagnosis in neonatea) | 113/135 (85.6) | |||
| Prenatal US | 113 | |||
| Prenatal MRI | 3 (2.2) | |||
| In-utero procedure | 11/135 (8.1) | |||
| RFA | 8 | |||
| Aspiration | 2 | |||
| Cystic-amniotic shunt | 1 | |||
| Diagnostic work-up after deliverya) | (n=182) | (n=132) | (n=26) | |
| US | 105 (57.7) | 83 (62.9) | 10 (38.5) | |
| CT | 16 (8.8) | 9 (6.8) | 5 (19.2) | |
| MRI | 159 (87.4) | 112 (84.8) | 24 (92.3) | |
| Preoperative biopsy | 4 (2.2) | 0 | 4 (15.4) | |
| Other | 3 (1.6) | 2 (1.5) | 1 (3.8) | |
| Pre-op AFP (ng/mL) | (n=138) | (n=111) | (n=14) | |
| Mean±SD | 87,658±77,076 | 20,287±42,396 | ||
| Median (range) | 64,745 (0.8–600,000) | 2.1 (0.8–150,730) | ||
| Preoperative treatment | (n=189) | (n=137) | (n=27) | |
| Preoperative chemotherapy | 6 | 6 | ||
| Preoperative embolization | 1 | 1 | ||
| Preoperative chemotherapy and ASCTb) | 1 | 1 | ||
| Preoperative complications | (n=189) | (n=137) | (n=27) | |
| Yes | 30 (15.9) | 26 (19.0) | 1 (3.7) | |
| No | 159 (84.1) | 111 (81.0) | 26 (96.3) | |
Values are presented as number (%).
US, ultrasound; MRI, magnetic resonance imaging; RFA, radiofrequency ablation; CT, computed tomography; AFP, α-fetoprotein; SD, standard deviation; ASCT, autologous stem cell transplantation.
a)Including multiple selection; b)Autologous stem cell transplantation.
| Characteristic | Total (n=189) | Neonate (n=137) | Old-age (n=27) | |
|---|---|---|---|---|
| Age at time of surgery (day) | ||||
| Mean | 305.9±957.1 | 6.9±9.2 | 1,985.3±1,784.9 | |
| Median | 6 (0–7,217) | 4 (0–63) | 1,426 (397–7,217) | |
| No. of operations | ||||
| 1 | 145 (76.7) | 106 (77.4) | 21 (77.8) | |
| 2 | 37 (19.6) | 27 (19.7) | 3 (11.1) | |
| 3 | 6 (3.2) | 4 (2.9) | 2 (7.4) | |
| 5 | 1 (0.5) | 0 | 1 (3.7) | |
| Body weight at time of surgery (kg) | ||||
| Mean±SD | 6.2±8.8 | 3.1±0.6 | 22.0±15.9 | |
| Median (range) | 3 (2–69) | 3 (2–5) | 16 (8–69) | |
| Operation time | ||||
| Mean±SD | 148.0±89.6 | 150.3±82.8 | 155.7±129.4 | |
| Median (range) | 125 (20–590) | 132 (20–410) | 110 (34–590) | |
| Mode of surgery | ||||
| Perineal approach | 177 (93.7) | 131 (95.6) | 23 (85.2) | |
| Perineal+laparotomy/laparoscopic | 9 | 5 | 2 | |
| Others | 3 | 1 | 2 | |
| Results of operation | ||||
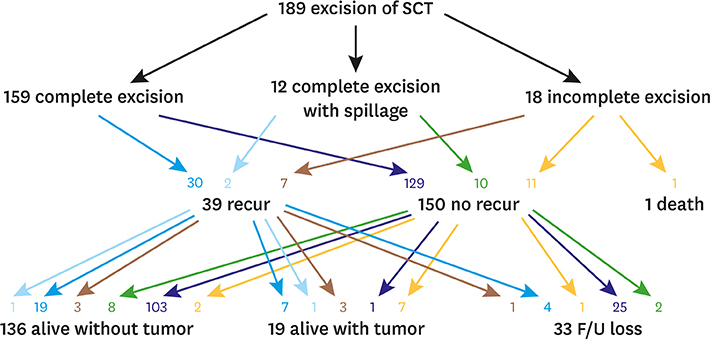
| Complete excision | 159 (84.1) | 116 (84.7) | 23 (85.2) | |
| Complete excision with spillage | 12 (6.3) | 6 (4.4) | 2 (7.4) | |
| Incomplete excision | 18 (9.5) | 15 (10.9) | 2 (7.4) | |
| Intraoperative complications | 21/189 (11.1) | 15/137 (10.9) | 3/27 (11.1) | |
| Bleeding | 8 (38.1) | 6 (40.0) | 2 (66.7) | |
| CSF leakage | 4 (19.0) | 1 (6.7) | 1 (33.3) | |
| CPCR | 4 (19.0) | 4 (26.7) | 0 | |
| Other complications | 6 (28.6) | 5 (33.3) | 0 | |
| Postoperative complicationsa) | 30/189 | 24/137 | 3/27 | |
| Bleeding | 3 (10.0) | 3 (12.5) | 0 (0) | |
| Wound problem | 17 (56.7) | 14 (58.3) | 1 (33.3) | |
| Intestinal obstruction | 1 (3.3) | 1 (4.2) | 0 (0) | |
| DIC | 5 (16.7) | 5 (20.8) | 0 (0) | |
| Other | 7 (23.3) | 4 (16.7) | 2 (66.7) | |
| Reoperation during hospitalization | 9/189 (4.8) | 7/137 (5.1) | 1/27 (3.7) | |
| Postoperative chemotherapy | 11 (5.8) | 5 (3.6) | 4 (14.8) | |
Values are presented as number (%).
SD, standard deviation; CSF, cerebrospinal fluid; CPCR, cerebral resuscitation; DIC, disseminated intravascular coagulation.
a)Including multiple selection.
| Characteristic | Total (n=189) | Neonate (n=137) | Old-age (n=27) | ||
|---|---|---|---|---|---|
| Pathology of tumor | |||||
| Mature teratoma | 138 (73.0) | 103 (75.2) | 16 (59.3) | ||
| Immature teratoma | 33 (17.5) | 30 (21.9) | 2 (7.4) | ||
| Grade 1 | 4 (13.8) | 4 (14.8) | 0 (0) | ||
| Grade 2 | 10 (34.5) | 8 (29.6) | 1 (100) | ||
| Grade 3 | 15 (51.7) | 15 (55.6) | 0 (0) | ||
| Grade unknown | 4 (13.8) | 3 | 1 | ||
| Mixed | 4 (2.1) | 2 (1.5) | 0 | ||
| Malignant (yolk sac) | 2 (1.1) | 1 (0.7) | 1 (3.7) | ||
| Othera) | 12 (6.3) | 1 (0.7) | 8 (29.6) | ||
| Largest tumor length (cm) | 6.8±4.8 | 7.9±5.1 | 4.2±4.8 | ||
| Type of tumor component | |||||
| Cystic type | 63 (34.1) | 43 (31.6) | 9 (37.5) | ||
| Predominantly cystic mixed type | 61 (33.0) | 48 (35.3) | 6 (25.0) | ||
| Predominantly solid mixed type | 32 (17.3) | 29 (21.3) | 1 (4.2) | ||
| Solid type | 29 (15.7) | 16 (11.8) | 8 (33.3) | ||
| Unknown | 4 | 1 | 2 | ||
| Altman classification | |||||
| I | 68 (36.0) | 53 (38.7) | 8 (29.6) | ||
| II | 51 (27.0) | 47 (34.3) | 2 (7.4) | ||
| III | 25 (13.2) | 21 (15.3) | 1 (3.7) | ||
| IV | 45 (23.8) | 16 (11.7) | 16 (59.3) | ||
Values are presented as number (%) or mean±standard deviation.
a)Epidermal cyst (2), lipoma (2), lipoblastoma (2), duplication cyst (1), solitary fibrous tumor (1), infantile fibrosarcoma (1), lipomyelomeningocele (1), lymphangioma (1), epithelioid hemangioendothelioma (1).
| Characteristic | Total (n=189) | Neonate (n=137) | Old-age (n=27) | |
|---|---|---|---|---|
| Postoperative follow-up (mo) | ||||
| Age at last follow-up | 47.9±38.9 (median 41, range 0–243) | |||
| Postoperative follow-up | 37.9±27.6 (median 34, range 0–112) | |||
| Follow-up methoda) | ||||
| US | 89 (47.1) | 66 (48.2) | 13 (48.1) | |
| CT | 23 (12.2) | 17 (12.4) | 4 (14.8) | |
| MRI | 98 (51.9) | 77 (56.2) | 10 (37.3) | |
| PET | 5 (2.6) | 3 (2.2) | 2 (7.4) | |
| Other | 4 (2.1) | 3 (2.2) | 0 (0) | |
| Follow-up interval (mo) | ||||
| 1–3 | 25 | |||
| 4–6 | 30 | |||
| 7–12 | 37 | |||
| >12 | 14 | |||
| Tumor recurrence | 39 (20.6) | 28 | 5 | |
| Detection of tumor recurrence | ||||
| Physical examination | 1 (2.6) | 1 (3.6) | 0 (0) | |
| Elevated tumor marker | 3 (7.7) | 3 (10.7) | 0 (0) | |
| MRI | 27 (69.2) | 20 (71.4) | 5 (100) | |
| U/S | 7 (17.9) | 4 (14.3) | 0 (0) | |
| Other | 1 (2.6) | 0 (0) | 0 (0) | |
| Treatment of tumor recurrence | ||||
| Excision | 21 (53.8) | 14 (50.0) | 3 (60.0) | |
| Excision+CTx | 12 (30.8) | 9 (32.1) | 1 (20.0) | |
| CTx only | 2 (5.1) | 1 (3.6) | 1 (20.0) | |
| Observation | 2 (5.1) | 2 (7.1) | 0 (0) | |
| Other | 2 (5.1) | 2 (7.1) | 0 (0) | |
| Long-term functional complicationsa) | 44 | |||
| Constipation | 21 | |||
| Soiling | 14 | |||
| Urinary incontinence | 7 | |||
| Lower extremity weakness | 5 | |||
| Other | 7 | |||
Values are presented as mean±standard deviation or number (%).
US, ultrasound; CT, computed tomography; MRI, magnetic resonance imaging; PET, positron emission tomography; CTx, chemotherapy.
a)Including multiple selection.
| Characteristic | Total (n=33) | Neonate (n=23) | Old-age (n=4) | |
|---|---|---|---|---|
| Mature | 19 (57.6) | 15 (78.9) | 1 (25.0) | |
| Original pathology | Mature (10), immature (8), mixed (1) | |||
| Immature | 2 (6.1) | 2 (8.7) | 0 (0) | |
| Original pathology | Mature (1), immature (1) | |||
| Malignant (yolk sac) | 6 (18.2) | 3 (13.0) | 1 (25.0) | |
| Original pathology | Mature (3), immature (1), yolk sac (2) | |||
| Mixed | 1 (3.0) | 1 (4.3) | 0 (0) | |
| Original pathology | Mature (1) | |||
| Othera) | 5 (15.2) | 2 (8.7) | 2 (50.0) | |
| Original pathology | Mature (3), immature (1), epithelioid hemangioendothelioma (1) | |||
a)Epithelioid hemangioendothelioma (1), inflamed granulation tissue (1), lipomeningomyelocele (1), lipoma (1), no evidence of residual teratoma but r/o recurred tumor on follow-up magnetic resonance imaging (1).
| Questionnaire | No. | |
|---|---|---|
| 1. Which of the following tests are the most important for the preoperative diagnosis of sacrococcygeal teratoma (excluding physical findings and multiple selections available)? | ||
| ① AFP | 12 | |
| ② US | 4 | |
| ③ CT | 6 | |
| ④ MRI | 16 | |
| ⑤ Biopsy | 0 | |
| 2. Have you needed to do a coccyx resection for patients with sacrococcygeal teratoma? please describe the method and extent of resection. | ||
| ① No resection | 0 | |
| ② Resection | 22 | |
| Method: electrical cautery and en bloc resection | ||
| 3. Who do patients follow-up with after surgery? | ||
| ① Pediatric surgeon | 14 | |
| ② Pediatrician | 4 | |
| ③ Other—both pediatric surgeons and pediatricians (especially in cases of immature or malignant pathologies) | 4 | |
| 4. What tests are performed after surgery? (Multiple options are possible.) | ||
| ① AFP | 21 | |
| ② US | 11 | |
| ③ CT | 4 | |
| ④ MRI | 12 | |
| ⑤ Other—rectal exam | 1 | |
| 5. What is the timing (interval) of the postoperative follow-up? | ||
| ① Every 6 months after surgery | 9 | |
| ② Every year after surgery | 7 | |
| ③ Every 2 years after surgery | 0 | |
| ④ Less than every 6 months after surgery | 4 | |
| 6. How long after surgery do you follow-up? (n=20) | ||
| ① Until 1 year after surgery | 0 | |
| ② Until 2 years after surgery | 1 | |
| ③ Until 3 years after surgery | 3 | |
| ④ Until 4 years after surgery | 0 | |
| ⑤ Until 5 years after surgery | 13 | |
| ⑥ Mature-3 years, immature-5 years | 3 | |
| 7. Do you have any experience with minimally invasive surgery for sacrococcygeal teratoma? | ||
| ① Yes | 5 | |
| ② No | 17 | |
AFP, α-fetoprotein; US, ultrasound; CT, computed tomography; MRI, magnetic resonance imaging.
a)These studies were published in Advances in Pediatric Surgery.
Values are presented as mean±standard deviation or number (%).
a)Including multiple selection.
Values are presented as number (%).
US, ultrasound; MRI, magnetic resonance imaging; RFA, radiofrequency ablation; CT, computed tomography; AFP, α-fetoprotein; SD, standard deviation; ASCT, autologous stem cell transplantation.
a)Including multiple selection; b)Autologous stem cell transplantation.
Values are presented as number (%).
SD, standard deviation; CSF, cerebrospinal fluid; CPCR, cerebral resuscitation; DIC, disseminated intravascular coagulation.
a)Including multiple selection.
Values are presented as number (%) or mean±standard deviation.
a)Epidermal cyst (2), lipoma (2), lipoblastoma (2), duplication cyst (1), solitary fibrous tumor (1), infantile fibrosarcoma (1), lipomyelomeningocele (1), lymphangioma (1), epithelioid hemangioendothelioma (1).
Values are presented as mean±standard deviation or number (%).
US, ultrasound; CT, computed tomography; MRI, magnetic resonance imaging; PET, positron emission tomography; CTx, chemotherapy.
a)Including multiple selection.
a)Epithelioid hemangioendothelioma (1), inflamed granulation tissue (1), lipomeningomyelocele (1), lipoma (1), no evidence of residual teratoma but r/o recurred tumor on follow-up magnetic resonance imaging (1).
AFP, α-fetoprotein; US, ultrasound; CT, computed tomography; MRI, magnetic resonance imaging.